CLINICALLY PROVEN
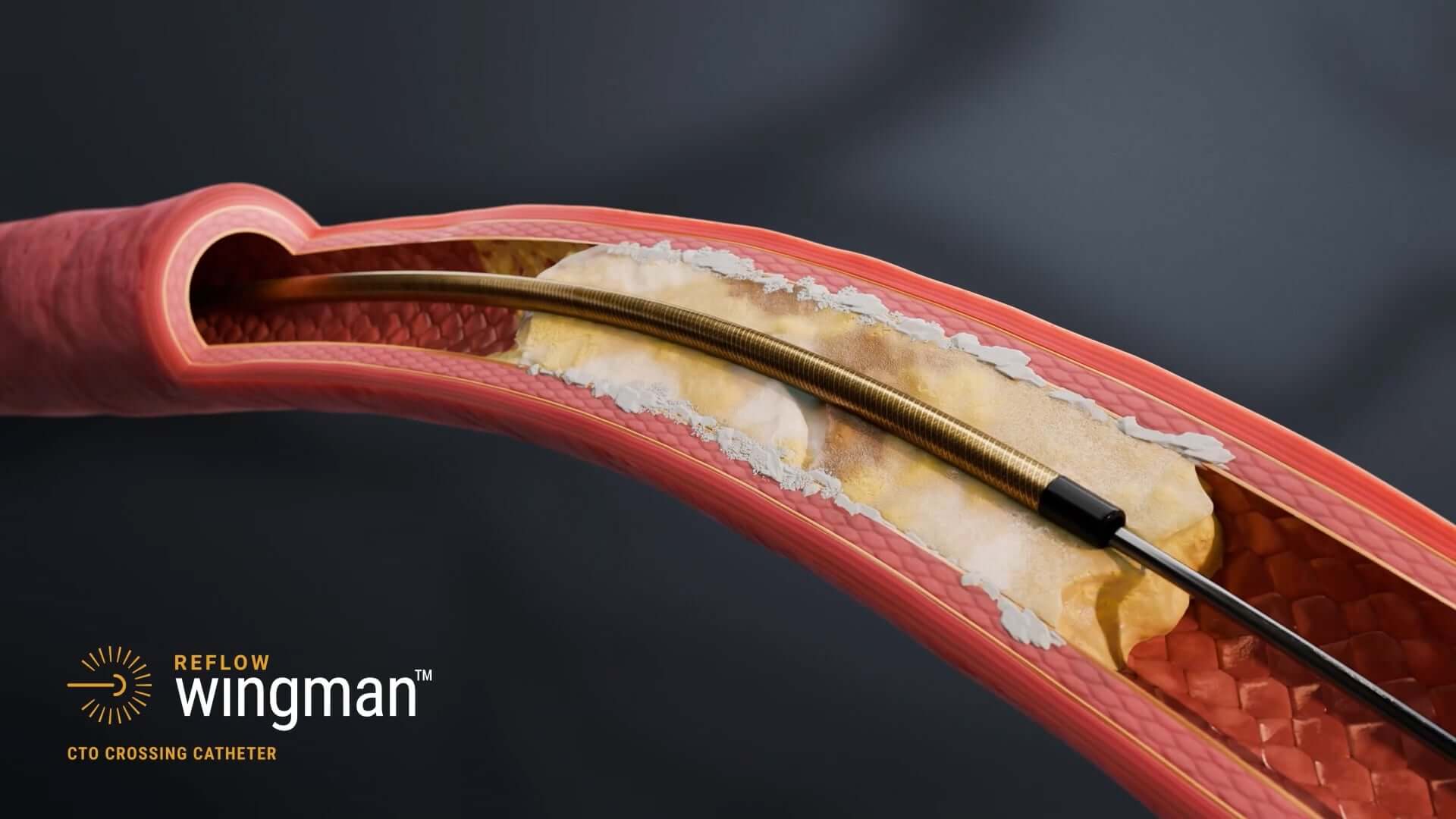
Physician Controlled Beveled Needle Tip

UNIQUE EXTENDABLE AND
ROTATING BEVELED
NEEDLE TIP
Simplifies entry into proximal cap and enables intraluminal CTO crossing
INTUITIVE
EASY-TO-USE
HANDLE
Allows you to simultaneously “push and twist” the beveled tip to anchor and penetrate the lesion
HIGH CROSSING SUCCESS RATE
AND SHORT CROSSING TIME
Clinically Proven Efficiency in the Wing-It Clinical Trial2

BRAID-REINFORCED
CATHETER SHAFT
Provides added strength for pushability through difficult lesions and kink resistance

HYDROPHILIC
COATING
Increases lubricity without compromising tactile feedback while enhancing trackability1

TELESCOPING
COMPATIBILITY
Wingman 14/14c allow telescoping with Spex LP 35 and Spex 35 Shapeable Support Microcatheters for angulation, additional support, or coaxial alignment
References
1. Wingman 14 is not hydrophilically coated.
2. Laird JR, Mathews SJ, Brodmann M, Soukas PA, Schmidt A, The Wing-It Trial Investigators. Performance of the Wingman catheter in peripheral artery chronic total occlusions: Short-term results from the international Wing-It trial. Catheter Cardiovasc Interv. 2021;97:310–316. doi:10.1002/ccd.29366
©2026 Reflow Medical, Inc. All rights reserved. Reflow Medical, Wingman and The Pulse of Medical Ingenuity are registered trademarks or trademarks of Reflow Medical, Inc. This device is restricted to use by or on the order of a physician. Refer to the Instructions for Use for a complete listing of the Indications, Contraindications, Warnings, Precautions, Adverse Events, and Procedural Steps.
Indications, Safety, & Warnings
CAUTION: This device is restricted to use by or on the order of a physician. Refer to the Instructions for Use for a complete listing of the Indications, Contraindications, Warnings, Precautions, Adverse Events, and Procedural Steps.
Indications for Use
The Wingman CTO Crossing Catheter is intended to treat peripheral artery disease (PAD) and chronic total occlusions (CTO).
Intended Use
The Wingman CTO Crossing Catheter is intended to be used in conjunction with steerable guidewires to access discrete regions of the peripheral vasculature and to penetrate and recanalize a lesion intraluminal or subintimal using the extendable needle tip with the activating handle. It may be used to facilitate placement and exchange of guidewires and other interventional devices and provide a conduit for delivery of saline solutions or diagnostic contrast.
Contraindications
The Wingman 14, 14C, 18, and 35 Crossing Catheters are contraindicated for use in the coronary and cerebral vasculature.
Warnings
• Hydrophilic wires prone to excessive swelling (e.g. ZipWire) should not be used with the Wingman CTO Crossing Catheter.
• Single Use only. Do not reuse/resterilize. Reusing the device could result in compromised device performance, cross-infection and other safety related hazards including patient injury.
• Do not use if device is open or packaging is damaged
• Never advance, withdraw or rotate the Wingman CTO Crossing Catheter against resistance until the cause is determined by fluoroscopy.
• DO NOT turn the handle more than 90 degrees during advancement of the extendable needle tip. The tip will not advance further by turning more than 90 degrees; continuing to rotate the handle beyond 90 degrees may lead to device breakage.
• This device contains nickel and should not be used in patients with known allergies to nickel.
• If the catheter is damaged, this product may cut into a blood vessel wall. Extreme caution needs to be taken when removing a damaged device.
• In the case of complications resulting from the removal of the entire system, stop immediately the procedure, and perform appropriate treatment at the discretion of the physician.
• This device contains cobalt and should not be used in patients with known allergies to cobalt.
Precautions
• Store in a cool, dry, dark place. Storage of the device in extreme conditions may damage the device and/or affect device performance that could lead to patient injury.
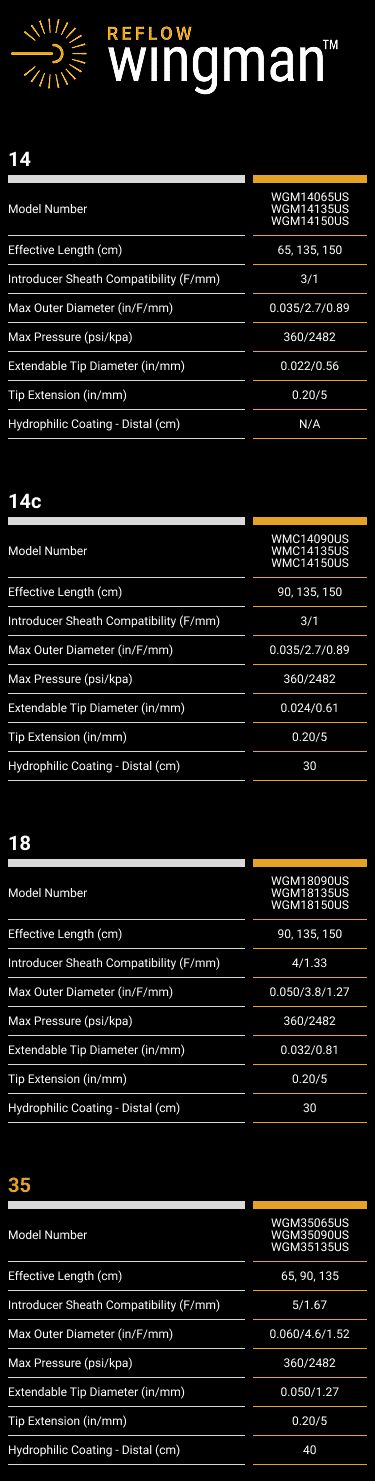
• Use only appropriately sized ancillary device, as shown in the Specifications above.
• Maximum Injection pressure: 360 psi (2482kpa).
• Use the catheter prior to the “Use By” date specified on the package.
• The catheter should only be used by physicians qualified to perform percutaneous vascular interventions.
• Precautions to prevent or reduce clotting should be taken when any catheter is used in the vascular system. Use of systemic heparinization and heparinized saline solution should be considered.
• Exercise care while handling the catheter during procedure to reduce the possibly of accidental damage, kinking or bending.
• Manipulation of the catheter should only occur under fluoroscopy.
©2026 Reflow Medical, Inc. All rights reserved. Reflow Medical, Wingman and The Pulse of Medical Ingenuity are registered trademarks or trademarks of Reflow Medical, Inc. This device is restricted to use by or on the order of a physician. Refer to the Instructions for Use for a complete listing of the Indications, Contraindications, Warnings, Precautions, Adverse Events, and Procedural Steps.

QUESTIONS?
TALK TO A REFLOW PRO.
For more information, please send us your question or request today.
A Reflow pro will get back to you.